![[Translate to Englisch:] Cartridge filling](/media/content/Bilder/Produktion/Cartridge-filling.jpg)
By 2050, it is projected that the proportion of people aged 65 and older will be more than double that of children aged 5. This global phenomenon is also leading to an increasing number of people suffering from chronic diseases such as diabetes and osteoporosis.
Combined, these trends are making it difficult for health care professionals to effectively treat patients over the long term. Thus, increased attention is leading to a growth in patient-friendly administration systems such as single-and dual-chamber cartridges in combination with a pen device.
For pharmaceutical and biotech companies looking to gain a competitive edge, pen injectors and cartridges offer an attractive alternative to vials. This combination features advanced design and offers product differentiation and improved life cycle management. But with these advantages come new challenges.
Current drivers of a cartridge-pen combination:
- Growing concern for patient safety, compliance, and consistency
- Increasing need toward patient self-medication at home
- Preferences for differentiation and greater ease of use
- Demand for self-administration in chronic diseases is rising
Different challenges with cartridge-pen combinations
Development of a cartridge-pen combination can be complex. To clarify, cartridge-pens or injector devices are considered combination products, which bring together a number of the following elements.
Combination products:
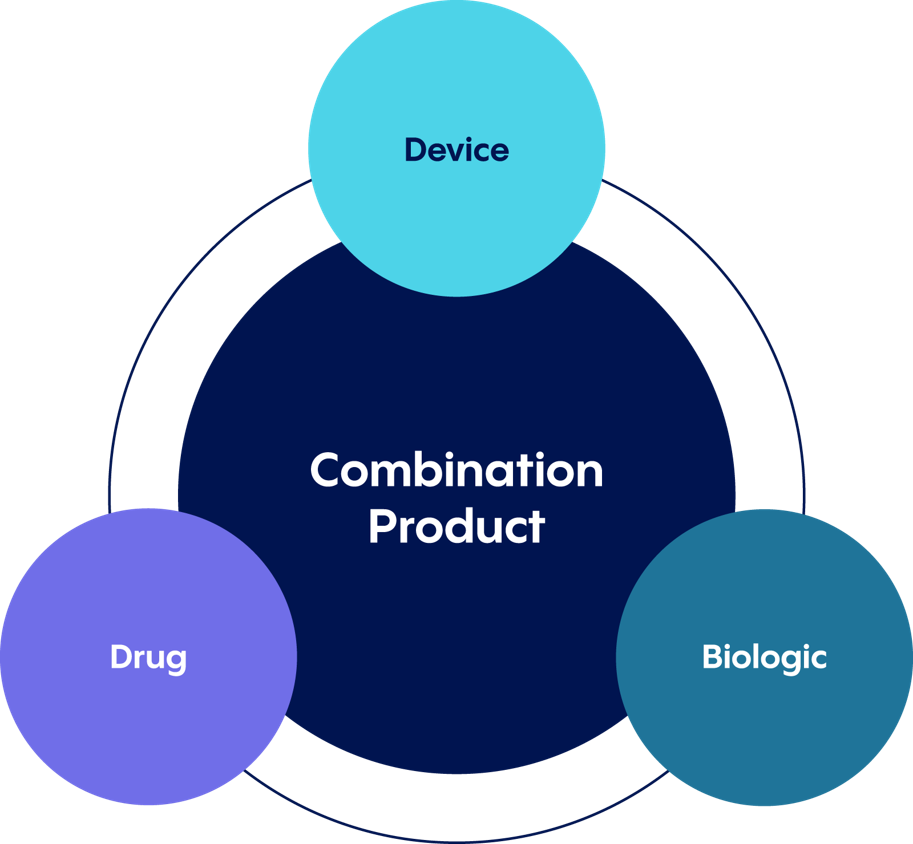
- A drug and a device
- A device and a biological product
- A biological product and a drug
- A drug, a device, and a biological product
Specific challenges with combination products include regulatory demands, current Good Manufacturing Practice (cGMP) requirements, and technical considerations.
Regulatory challenges:
Combination products are subject to specific FDA regulations related to their constituent parts, meaning components must be addressed individually. Human factor and clinical study considerations also need to be taken into account for combination products.
cGMP requirements:
Combination products must follow current GMP guidance, and as with regulatory demands, cGMP requirements apply to constituent parts.
Technical considerations:
Combination products require a special FDA marketing application. Following this guidance is key to a streamlined process.
Choosing the right partner for development
Partnering with a leader who is experienced in developing combination products can be invaluable when navigating special regulatory processes. Along with regulatory know-how, the following criteria should be considered when selecting a partner.
- A successful track record in cartridge development
- Capabilities in manufacturing processes
- Capabilities in cartridge filling – clinical and commercial
- Selection of packaging materials
- A manufacturing process that ensures tight product specifications
Additional considerations
Cartridges require a filling and pen assembly solution. A single point of contact can simplify accountability and response from your partner.
Look for a partner who offers a full range of services, customized packaging development, secondary packaging options, and expertise in complex packaging.
Vetter is your partner for cartridges
As a global expert, Vetter has a successful track record in bringing cartridges to market, with several cartridge products currently in development. With 40+ years of experience, Vetter understands the many challenges posed by combination products.
As one of only a small number of CDMOs able to develop assembly processes for pen devices, we provide a full scope of cartridge and pen platforms, including clinical and commercial offerings. Look to Vetter to make the most of your cartridge-pen combination product.


